Safer, simpler and efficient lab-scale method for generating important compounds
Skrydstrup Group has developed a simple, secure, and highly efficient protocol for producing aldehydes, which are important chemical building blocks. The method includes using their two-chamber reactor, and the results have been published in the highly renowned journal, Nature Catalysis. The synthetic methodology is foreseen to have an impact for drug discovery and development in both industry and academia.
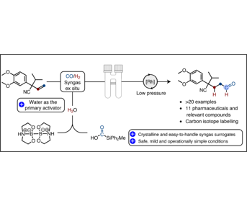
Aldehydes constitute a family of reactive, organic compounds that occur naturally in flavouring agents like benzaldehyde, which provides the odour and flavour of fresh almonds; cinnamaldehyde, or oil of cinnamon; and vanillin, the main flavouring agent of vanilla beans. Aldehydes can also be manufactured in the laboratory, and some aldehyde compounds are common building blocks for the production of other chemicals used in resins, perfumes, dyes, organic acids, solvents, or as intermediates in the manufacture of plastics, and pharmaceuticals.
Industrially, aldehydes are produced annually on a multi-million tonne scale via a process called hydroformylation of olefins with syngas, which is a mixture of carbon monoxide (CO) and hydrogen (H2). This application has not found frequent use for chemical synthesis in the laboratory. This can be ascribed to the necessary investment of costly infrastructure for the handling of extremely toxic and flammable gasses under high pressure and forcing reaction conditions that require sophisticated stainless-steel reactors in explosion secured facilities.
No need for expensive equipment
In a paper recently published in the highly ranked journal, Nature Catalysis, the iNANO-based Skrydstrup Group reports of a safer, simpler and highly efficient method, which may replace the more commonly used laborious techniques for the production of aldehydes in a laboratory setting.
The research team has developed an operationally simple and highly efficient protocol by using a two-chamber reactor previously developed by the Skrydstrup Group. In the two-chamber reactor the hydroformylation of primary alkenes operates efficiently under mild conditions, without the need for expensive equipment like gas cylinders and autoclaves. This methodology utilizes the synergistic combination of two easy-to-handle, easy-to-obtain and crystalline main element compounds, wherein water is the primary activator for syngas release.
The approach provides unique opportunities to access aldehydes in a safe and reliable manner with further adaptation to the synthesis of a range of pharmaceuticals and relevant molecules thereof. This strategy is adaptable to carbon isotope labelling as demonstrated by the use of a 13CO releasing molecule.
The hydroformylation approach is anticipated to provide a complementary toolbox for drug discovery and development in both industry and academia.
READ ALSO:
Read more about the results in Nature Catalysis:
Main element chemistry enables gas-cylinder-free hydroformylations by Samuel K. Pedersen, Haraldur G. Gudmundsson, Dennis U. Nielsen, Bjarke S. Donslund, Hans Christian D. Hammershøj, Kim Daasbjerg & Troels Skrydstrup
The research was carried out by researchers from Interdisciplinary Nanoscience Center (iNANO) and the Department of Chemistry at Aarhus University. The work was financially supported by Danish National Research Foundation and NordForsk.
For further information, please contact
Professor Troels Skrydstrup
Interdisciplinary Nanoscience Center and Department of Chemistry
Aarhus University
Email: ts@chem.au.dk